Molweight, melting and boiling point, density, flash point and autoignition temperature, as well as number of carbon and hydrogen atoms in each molecule for 200 different hydrocarbons. Variations in fuel oils density as function of temperatur, together with volume correction factors. Temperatureĭensity of Ethyl Alcohol aqueous solutions. Density of inorganic substances in water is plotted as function of wt%, mol/kg water and mol/l solution.Įthanol Water Mixtures - Densities vs. Density of some sugars, alcohols and other organic substances in water is plotted as function of wt%, mol/kg water and mol/l solution.ĭensity of Aqueous Solutions of some Inorganic SubstancesĬhanges in density of aqueous solutions with changes in concentration at 20☌. Online density converter with commonly used units.ĭensity of Aqueous Solutions of Organic Substances as Sugars and AlcoholsĬhanges in density of aqueous solutions with changes in concentration at 20☌. Density of acetic acid, citric acid, formic acid, D-lactic acid, oxalic acid and trichloroacetic acid in water is plotted as function of wt%, mol/kg water and mol/l solution. Density of inorganic sodium salts in water is plotted as function of wt%, mol/kg water and mol/l solution.ĭensities of Aqueous Solutions of Organic AcidsĬhanges in density of aqueous solutions with changes in concentration at 20☌. Density of potassium salts in water is plotted as function of wt%, mol/kg water and mol/l solution.ĭensities of Aqueous Solutions of Inorganic Sodium SaltsĬhanges in density of aqueous solutions with changes in concentration at 20☌. Density of inorganic chlorides in water is plotted as function of wt%, mol/kg water and mol/l solution.ĭensities of Aqueous Solutions of Inorganic Potassium SaltsĬhanges in density of aqueous solutions with changes in concentration at 20☌. Variations in crude oil density are shown as function of temperatur, together with volume correction factors.ĭensities of Aqueous Solutions of Inorganic ChloridesĬhanges in density of aqueous solutions with changes in concentration at 20☌. 
Online API to Specific Gravity calculator. 
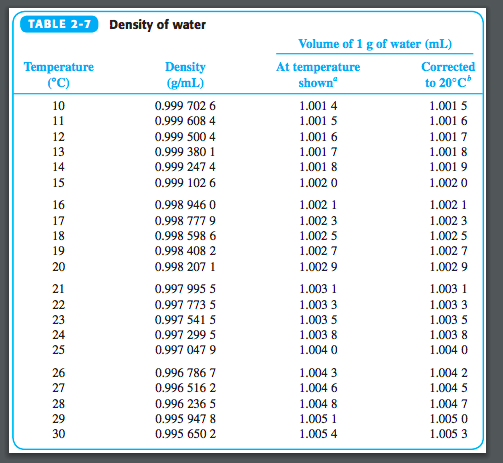
Molweight, melting and boiling point, density, pKa-values, as well as number of carbon and hydrogen atoms in molecules are given for 150 different alcohols and acids.ĪPI expresses the gravity or density of liquid petroleum products. Material properties of gases, fluids and solids - densities, specific heats, viscosities and more.Īlcohols and Carboxylic Acids - Physical Data Pipe lines - fluid flow and pressure loss - water, sewer, steel pipes, pvc pipes, copper tubes and more. You can divide pounds per cubic foot by 32.2 for a rough value in slugs.ĭensities of solids, liquids and gases. Note that even if pounds per cubic foot is often used as a measure of density in the U.S., pounds are really a measure of force, not mass. These variations of water changes its density because what's in the water has its own density.Ethyl Alcohol (Ethanol, pure alcohol, grain alcohol or drinking alcohol) Other factors affect water's density such as whether it is tap or fresh water or salt water. Water has the maximum density of 1 g/cm 3 only when it is pure water. When the temperature changes from either greater or less than 4 degrees, the density will become less then 1 g/cm 3. Water has its maximum density of 1g/cm 3 at 4 degrees Celsius. Water never has an absolute density because its density varies with temperature. The conversion between the two is 1000 kg/m 3 to 1 g/cm 3. The units used to indicate density are or more commonly used. The commonly used formula to determine the density of an object is ρ = m/V, ρ (rho) represents density, m represents mass, and V represents volume. Water turns into steam from 100 degrees and above.ĭensity is defined as mass per unit of volume. Water turns to ice as it's solid state from 0 degrees Celsius and below. Water is most common in it's liquid state when it is kept a normal pressure and between 0 degree Celsius and 100 degree Celsius. Water can change into three phases of matter. Almost 75% of the Earth's surface is covered with water and almost every living thing on Earth is made up of 90% water. Water is a very common substance here on Earth. Englewood Cliffs, NJ: Prentice Hall 1987: 72.ĭensity of Water, Simetric. Chemistry: The Study of Matter: Fourth Edition. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
